Terminology
First off, a little bit on the (potentially confusing) terminology: Since we all like acronyms in medicine you'll often hear any supraglottic airway referred to as an 'LMA', short for laryngeal mask airway, as in "... let's use an i-gel LMA".
These days 'LMA' is a registered trademark for a company that makes the LMA Unique® or LMA Supreme® for example. Therefore you should avoid the term 'LMA' unless you are talking about supraglottic airways made under this particular trademark. The correct umbrella term for all these devices is 'supraglottic airway' (SGA) or, also common, 'supraglottic airway device' (SAD).
Extraglottic airways
Another term you might come across is 'extraglottic airway'. These are devices that are positioned outside the glottis like SGAs but can also extend inferior/ caudad to it. Therefore all supraglottic airways are also extraglottic airways but some extraglottic airways are not supraglottic.
An example of an extraglottic (but not supraglottic) airway device is the Esophageal tracheal airway/ Combitube®.
History
The laryngeal mask was invented by Dr. Archibald ('Archie') Brain, a British anesthesiologist (or 'anaesthetist', to be precise) in the early 1980s and first came to market in the United Kingdom in 1987. Wikipedia has an excellent summary of how Dr. Brain developed the device.
Conincidentally, Propofol became widely available around the same time, which helped the laryngeal mask gain popularity quickly among practitioners. The reason for this is that Propofol works much better than other hypnotics, such as barbiturates, in suppressing laryngeal reflexes in non-paralyzed patients. It is therefore much easier to place a laryngeal mask after induction with Propofol than, for example, Thiopentone in patients who are not paralyzed and breathing spontaneously under general anesthesia.
The one supraglottic airway available today which most closely resembles the initial LMA design invented by Dr. Brain is the LMA Classic®, which is the re-usable version of the LMA Unique®.
FEATURES
All supraglottic airways generally have a tube component and a (more or less) antomically shaped cuff component. Beyond that, designs vary from one device to the next. The cuff is shaped in a way that helps the device 'sit' in its desired position with the opening of the cuff covering the laryngeal inlet.
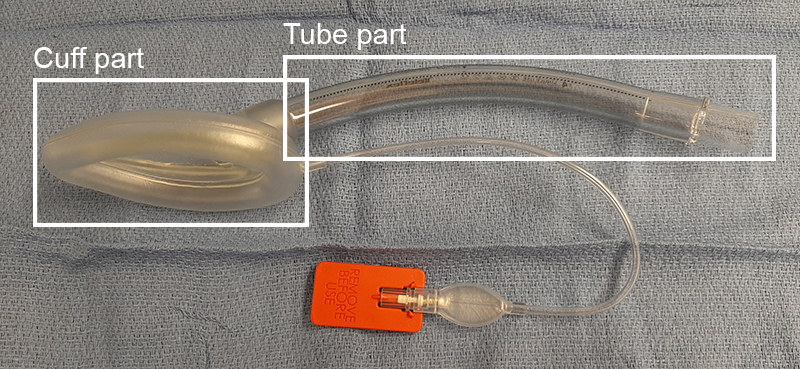
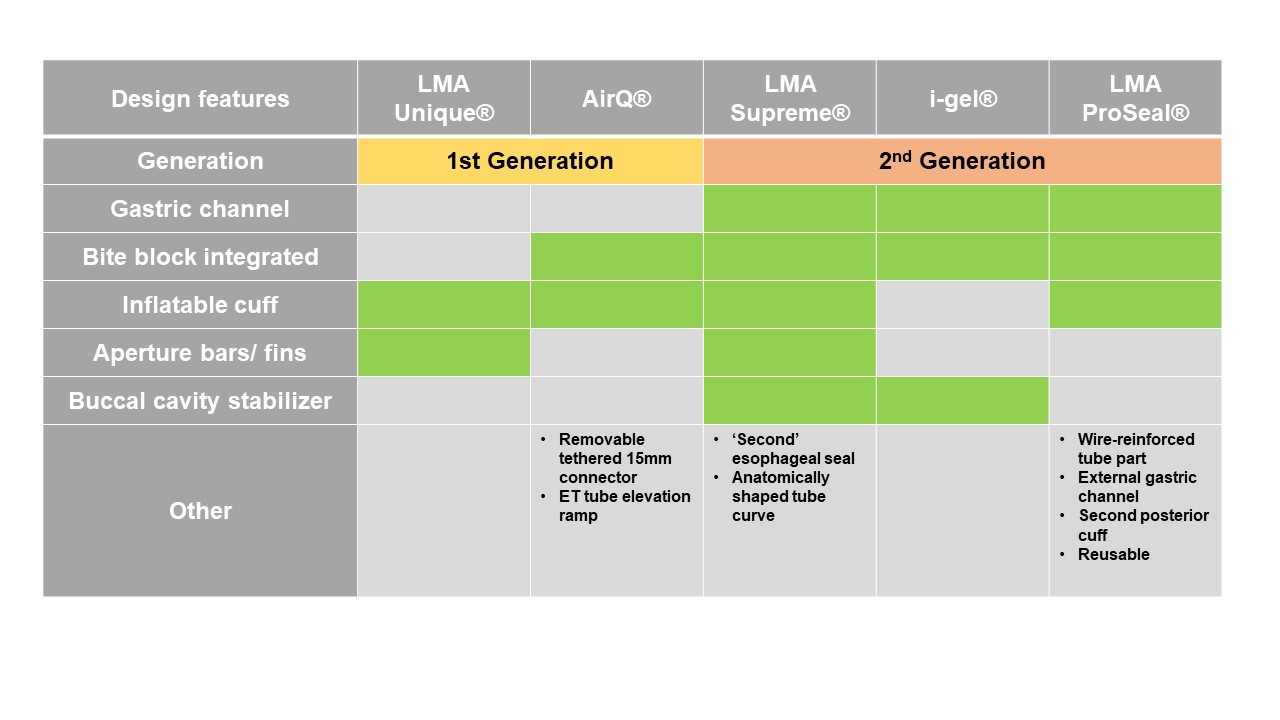
Many SGAs have an inflatable cuff with a pilot ballon whereas other don't. Some include a gastric channel, which is considered a design feature of second generation devices. Other SGAs may include a bite block or a buccal stabilizer. The variations are manifold given that there are over 20 types of SGAs currently being marketed.
'Efficacy' and 'safety' of supraglottic airways
SGAs can be described for their efficacy and their safety:
- Efficacy: This is about how well a device does what it is supposed to do, meaning how well the device works. For SGAs efficacy is determined by how well you can ventilate a patient through the device without a gas leak occuring around the cuff. The surrogate measurement for this is the cuff seal pressure, i.e. the airway pressure at which a cuff leak occurs.
- Safety: This is about to what degree a device doesn't do something you don't want it to do, meaning how well it avoids undesired effects. For SGAs safety relates to how well they avoid aspiration from happening.
With regards to SGAs their safety also depends somewhat on their efficacy. If you can ventilate through a particular device with comparatively high airway pressures without a leak occuring, meaning the cuff seal pressure is quite high, this device will also
a) prevent inadvertent gastric insufflation and regurgitation well, and
b) 'seal' the airway with the cuff preventing aspiration.
For SGAs therefore, better efficacy brings about better safety.
First and second generation devices
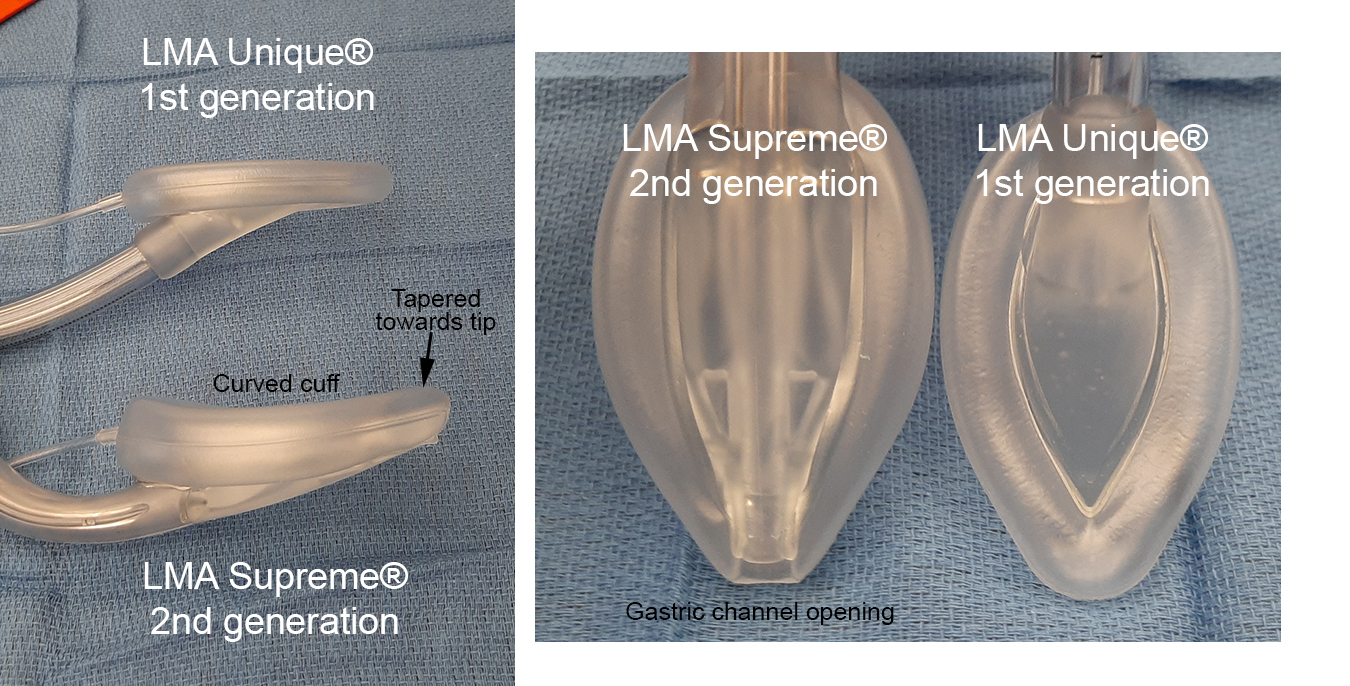
The most obvious example of a first generation SGA is the classic laryngeal mask design, today available as the LMA Unique® and LMA Classic® .
Second generation SGAs are defined as "those with specific design features intended to reduced the risk of aspiration", making these devices 'safer' than first generation ones. Reducing the risk of aspiration can be achieved two ways:
- Venting of regurgitant material: A very common and obvious feature of second generation devices is a gastric channel (also referred to as 'drain channel', 'drain tube', etc.). The purpose of this channel is, should regurgitation occur, to drain fluid away through the channel as a path of least resistance.
- Obturating the esophagus: The idea here is that the tip of the SGA cuff with its position below the laryngeal inlet and just above or inside the upper esophageal sphincter will occlude the esophagus and prevent regurgitant material from entering the hypopharynx. Second generation SGAs therefore have more evolved cuff tip designs to achieve better esophageal occlusion compared to first generation devices.
Second generation devices are also safer (reducing aspiration risk) by being more efficacious. Their cuffs are designed to achieve higher cuff seal pressures. As an example, the LMA Unique® has been found to achieve median cuff seal pressures of around 20 cmH2O, whereas the i-gel SGA seals to around 30 cmH2O.
CAUTION: It might be temping to use the gastric channel of second generation devices to place an esophageal temperature probe intra-operatively. If you do, you are disabling two crucial safety features of these devices: You are blocking the venting of regurgitant fluid and also potentially messing with the esophageal obturation the tip of the cuff is designed to achieve. Use a nasopharyngeal temperature probe instead.
Should first generation SGAs still be used?
Despite some shortcomings the classic laryngeal mask, today available as the LMA Classic® or LMA Unique®, is arguably the one SGA that has been investigated and tested the most. Many of the second generation SGAs marketed today on the basis of having superior efficacy and safety over the classic design have not been evaluated in clinical studies to the same extent, and some have not been tested in adequate clinical studies at all. Nevertheless, many argue that at least some of the second generation devices are so superior compared to first generation ones that we should abandon the 'vintage' SGAs and use of newer devices should be the norm (Cook & Kelly, 2015).
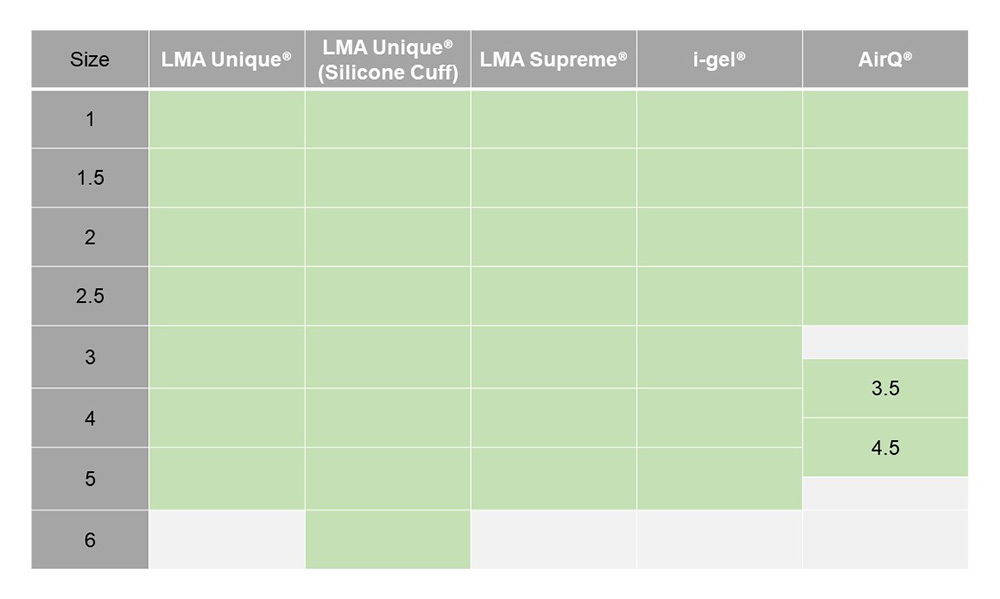
Sizes
SGAs come in a range of sizes and many devices are available in very small version suitable even for neonates. Sizing between different types of SGAs is roughly comparable but please refer to the links for individual devices to find more information on correct sizing. As a very crude guide, use a size 3 for small adults, size 4 for average size adults and most women, and size 5 for large adults and most men.
USES
Especially with the introduction of second generation devices the role of SGAs has expanded from airway maintenance during routine low-risk surgery. SGAs are now...
- increasingly used for more obese or 'higher risk' patients,
- used for as a conduit for intubation, and
- used for airway management outside of the OR, for example during resuscitation/ cardiac arrest or in pre-hospital emergency care, where they might offer higher ventilation success rates compared to orotracheal intubation, particularly when providers are not well-trained to intubate.
For a more detailed discussion on indications and contraindications for using SGAs, what patients and which types of surgery are suitable, please check out the page on 'SGA placement'.
